



1 / 5
 WER Medical
WER Medical










| Still deciding? Get samples of $ ! US$ 0/Piece Request Sample |
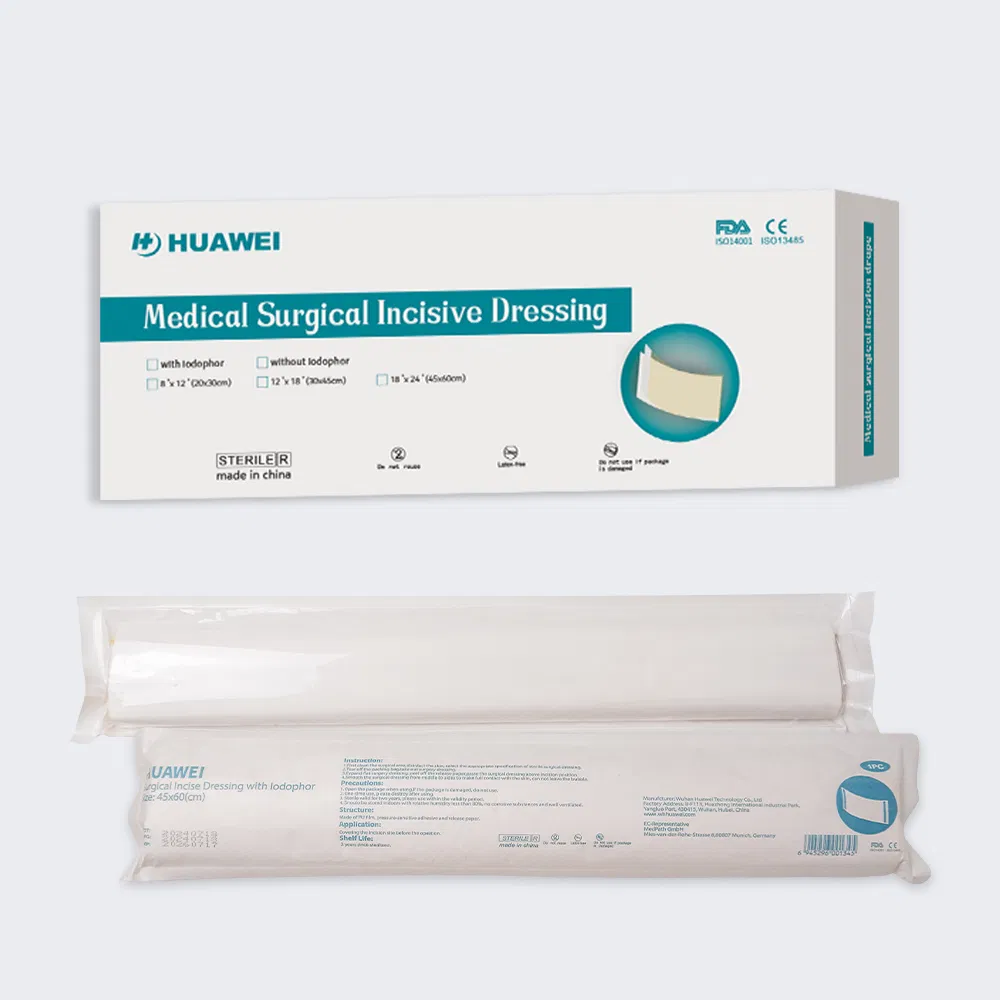
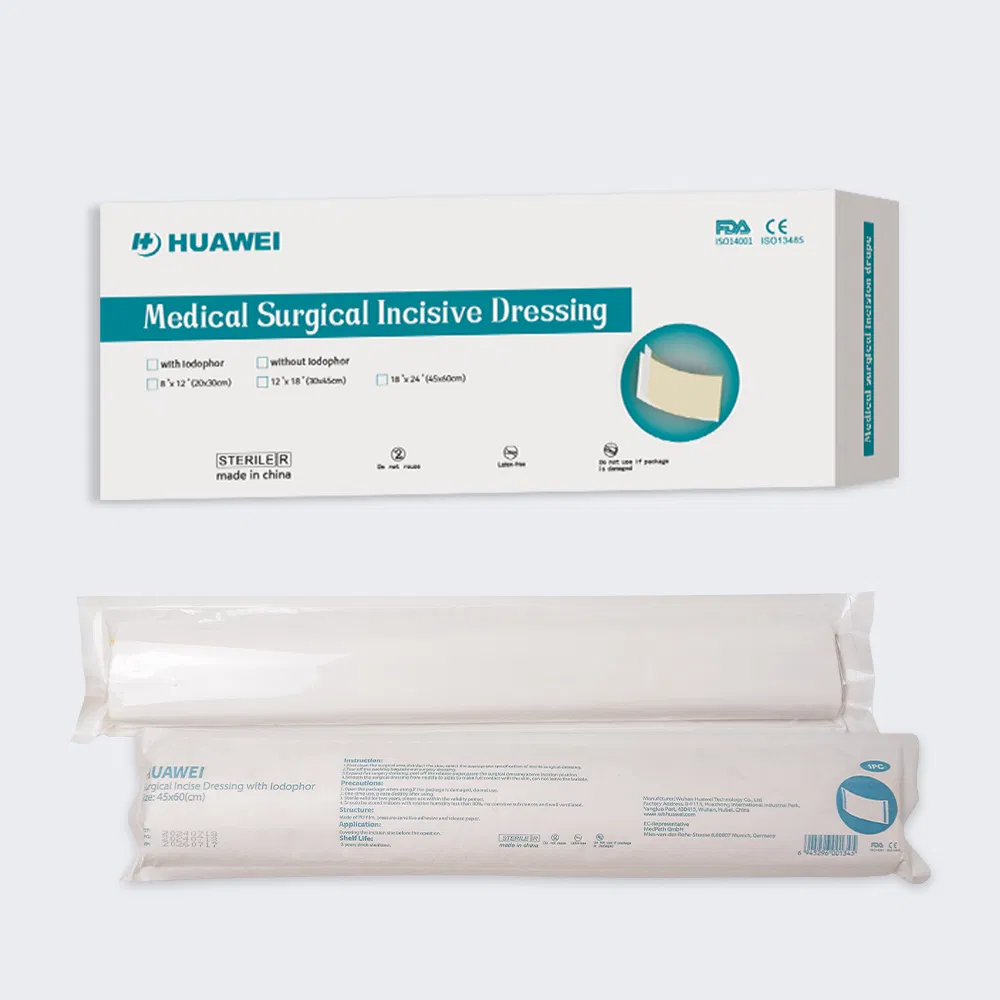
| Type | Name | Layers | Specification |
|---|---|---|---|
| 1 | PE surgical dressing | 3 layers | 0.02-0.05mm PE + adhesive + protection paper |
| 2 | PE surgical dressing with iodine | 3 layers | PE + adhesive with iodine + protection paper |
| 3 | PE surgical dressing (non-adhesive edges) | 3 layers | PE + iodine adhesive (two sides without adhesive) |
| 4 | PU surgical dressing with iodine | 3 layers | 0.02-0.05mm PU + iodine adhesive |
| 5 | PU surgical dressing | 4 layers | 0.05m PE + 0.02mm PU + adhesive + paper |
Size, shape, package, and printing can be fully customized to meet clinical requirements.





Our facility is a high-tech enterprise devoted to medical health products development, production, and sales. We adhere to "quality first, credibility supreme" as our business philosophy. With an independent and modernized R&D center, we have formed joint development systems with leading universities and international experts.
A key innovation is the breathable membrane waterborne polyurethane (WPU) imitation skin membrane. This domestic initiative product offers superior water resistance, anti-bacteria properties, and zero sensitization, exceeding traditional film materials.



