WER Medical
WER Medical










| Still deciding? Get samples of $ ! US$ 0.14/Piece Request Sample |
Professional Hydrocolloid Dressing
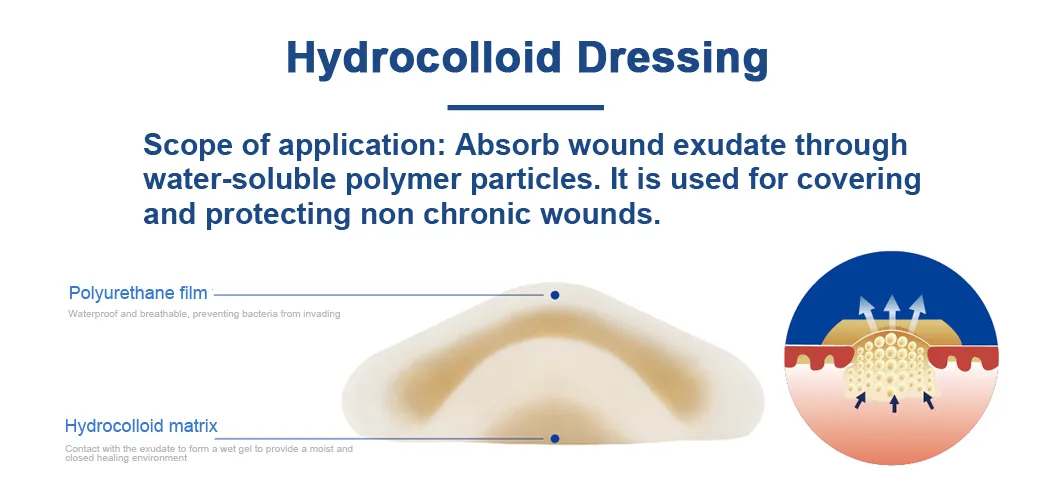
The hydrocolloid dressing combines the water absorption properties of the water-soluble polymer and the viscosity of the rubber material. The wet healing environment supplied by hydrocolloid dressing has a great effect on promoting wound healing, which can fully absorb wound exudate and prevent bacteria from invading wounds.
| Type | Code | Size (cm) | Pcs/Inner Box | Pcs/Master Carton |
|---|---|---|---|---|
| Thin | GL102 | 10*10 | 10 | 400 |
| GL104 | 15*15 | 10 | 400 | |
| Island | GL103 | 10*10 | 10 | 400 |
| GL107 | 15*15 | 10 | 400 |



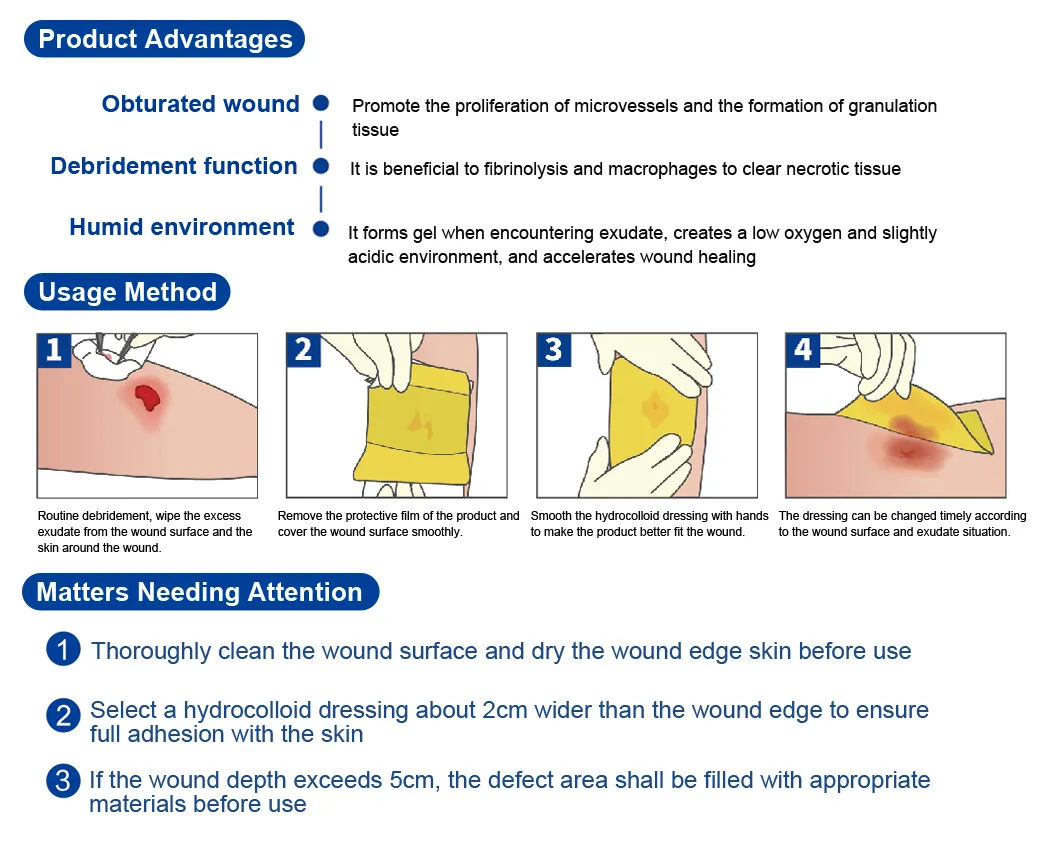
Promotes the proliferation of microvessels and the formation of granulation tissue for faster recovery.
Beneficial to fibrinolysis and macrophages to efficiently remove necrotic tissue from the wound site.
Forms a gel when encountering exudate, creating a low oxygen and slightly acidic environment to accelerate healing.
Founded in 2015, we are a professional medical technology enterprise specializing in the development, production, and sale of medical surface catheter fixation devices, transparent dressings, and specialized fixation bands. With advanced production equipment and a scientific management system, we hold production qualifications for Class I and Class II medical devices.