

 WER Medical
WER Medical










| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |
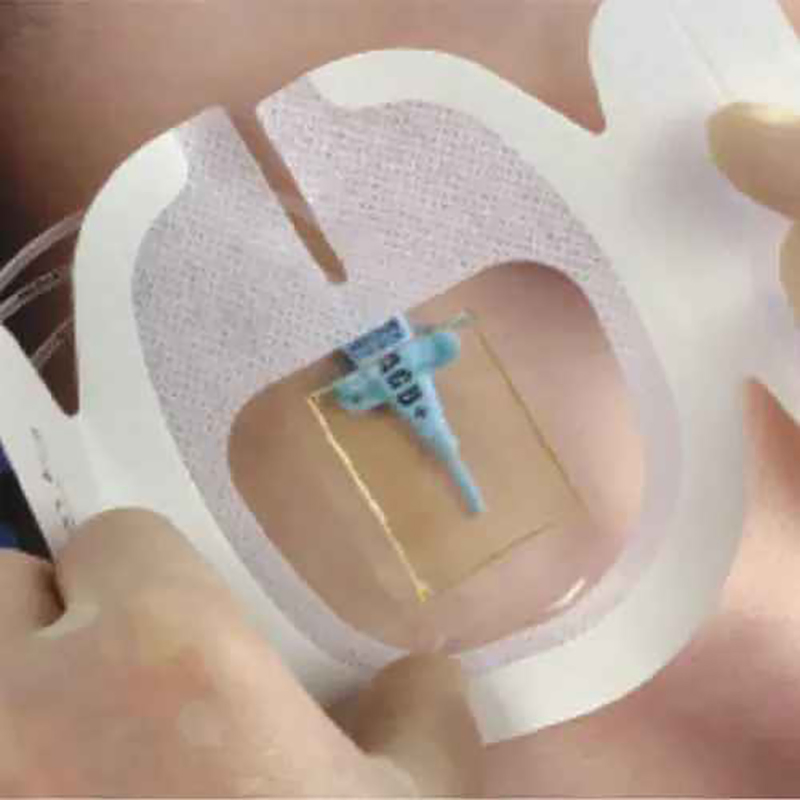
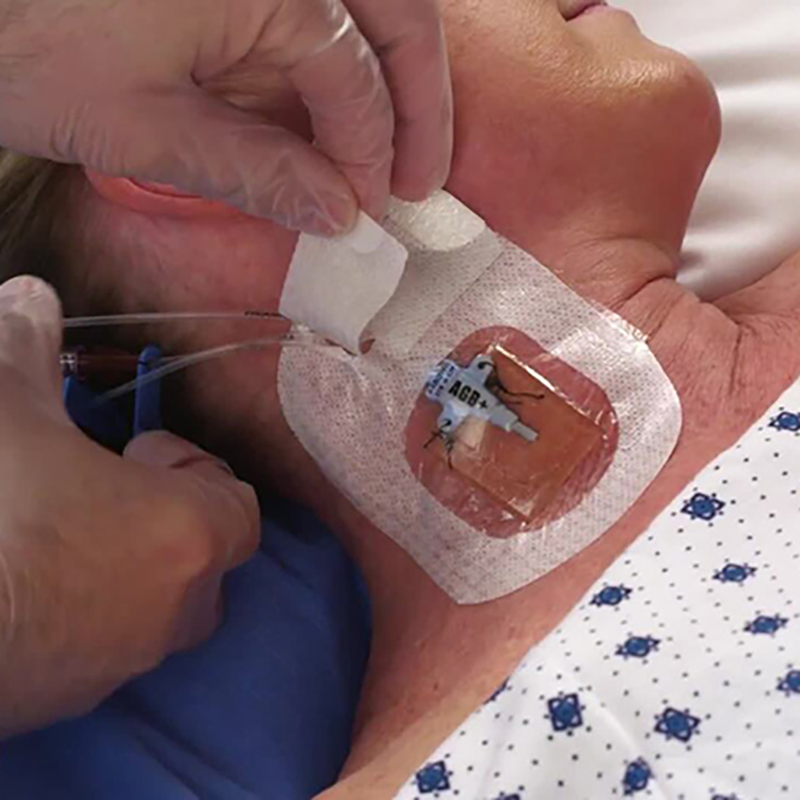
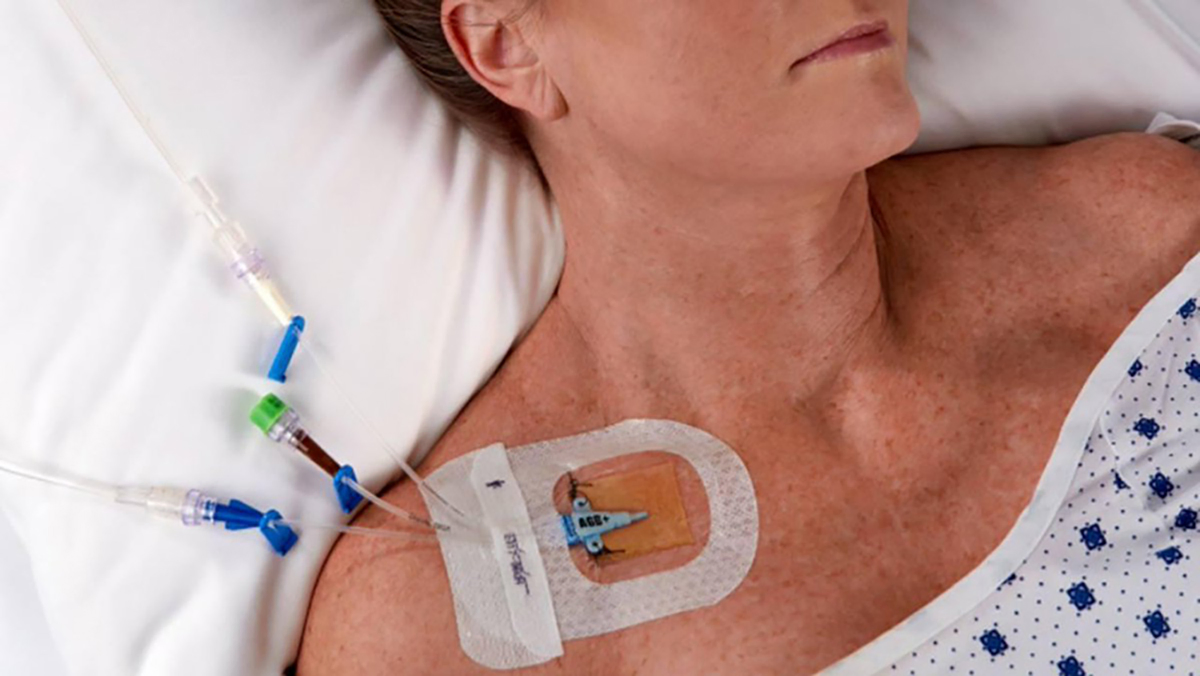
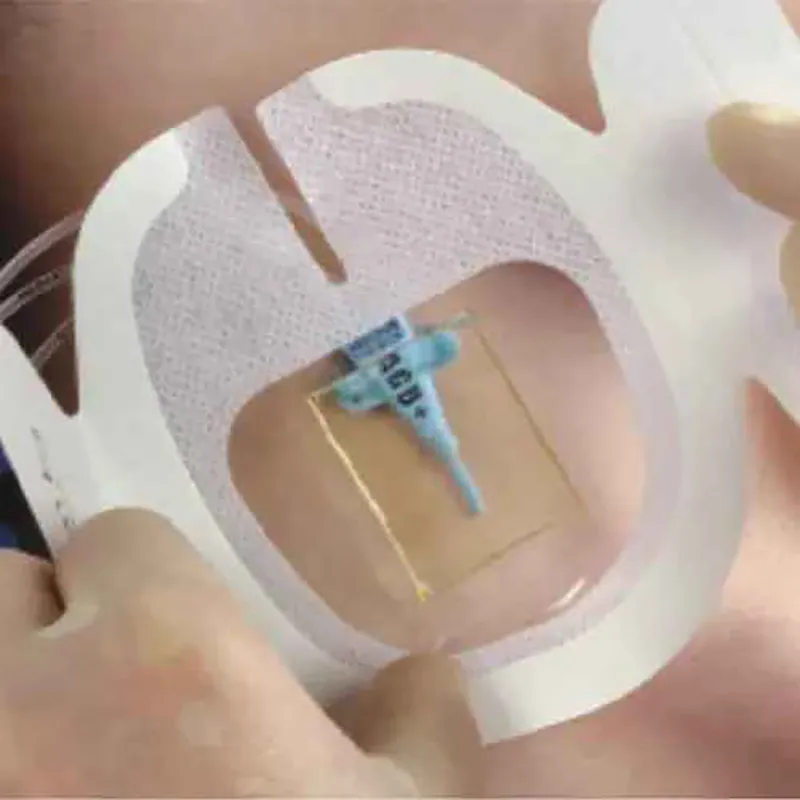
CHG IV Fixed Dressing
CHG I.V. dressing is composed of a transparent dressing and a gel pad. The gel pad contains 2% chlorhexidine gluconate (CHG), which is an antibacterial agent.
The dressing is used to fix and cover intravenous catheters, other intravascular catheters and percutaneous devices. It can also be used to reduce the number of skin colonies and inhibit the re-growth of microorganisms.
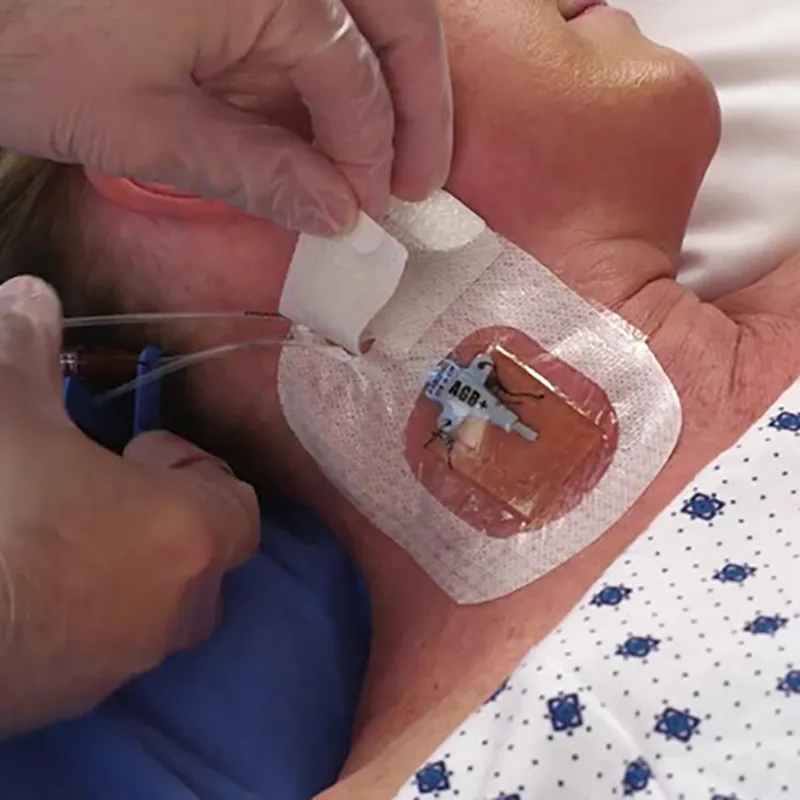
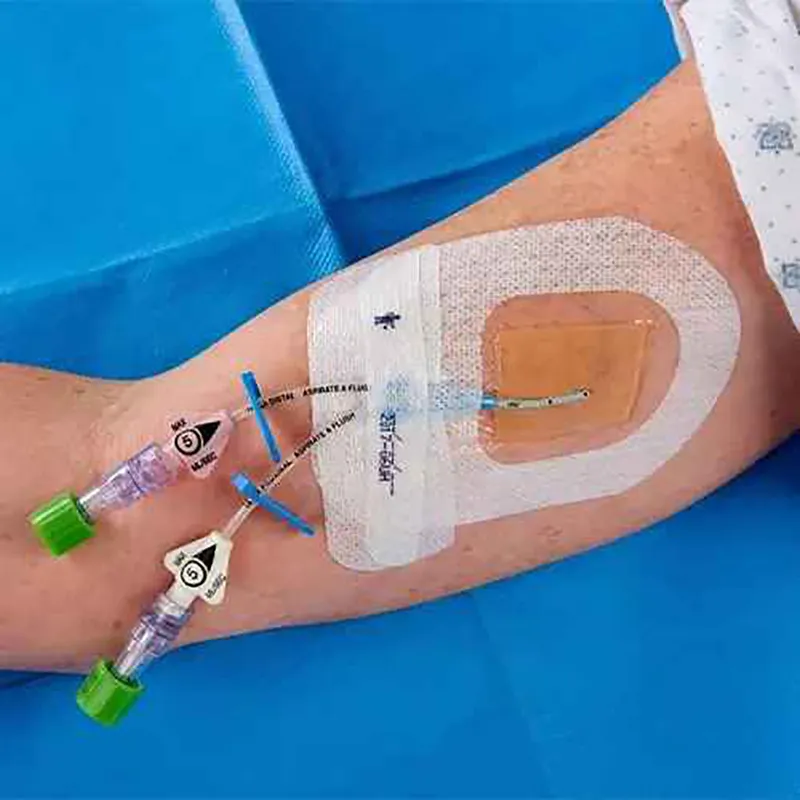
Key Advantages:
Size Specifications (cm):

We are a reliable long-term partner in the medical field. In 2020, our products were exported to 46 international countries and regions including North America, Europe, Southeast Asia, and the Middle East.
Our facility covers an area of 10,000 square meters, featuring a 5,000 square meter production workshop. Our infrastructure includes 294 sets of triply-wire machines, 120 sets of flat wire machines, 80 sets of welding machines, and 150 sets of mask machines. We operate two automatic warehouses with a storage capacity exceeding 43,000 cubic meters, supported by a 2,500 square meter ethylene oxide sterilization center capable of sterilizing 7,500 cubic meters per month.

