



 WER Medical
WER Medical










| Still deciding? Get samples of $ ! US$ 0.01/Piece Request Sample |
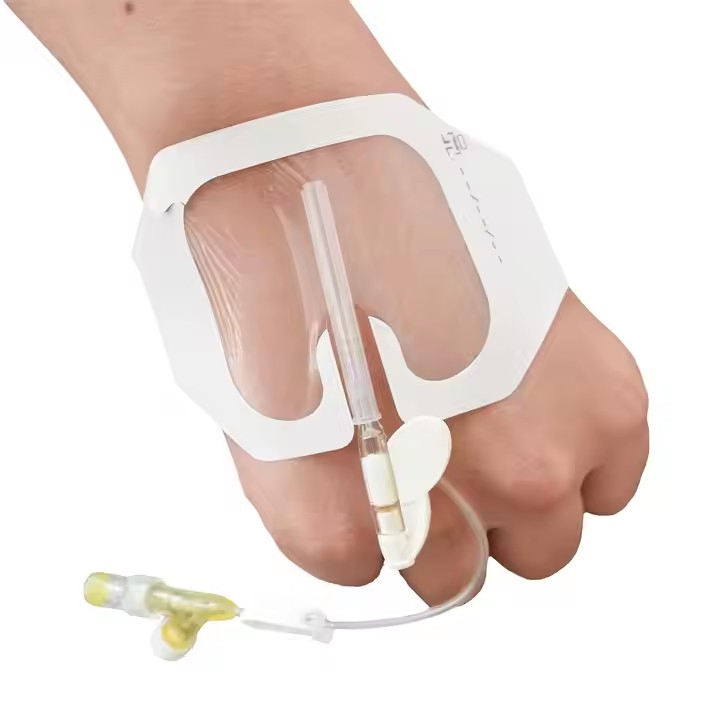
A sterile disposable IV cannula dressing is a medical device used to cover and protect an IV cannula insertion site. It is a sterile adhesive dressing that is placed over the site where the IV cannula is inserted into a patient's vein.
The dressing is typically made of a transparent film or breathable material that allows for visual inspection of the insertion site and allows air to reach the skin. It is designed to securely adhere to the skin and provide a barrier against moisture, bacteria, and other contaminants.
The dimensions of a typical sterile disposable IV cannula dressing are 6x8cm, but sizes may vary depending on the specific product requirements. The dressing is single-use and should be discarded after removal or if it becomes soiled.

| Items | Product Details | Package | Carton Size (CM) |
|---|---|---|---|
| TC1154 | IV CANNULA WOUND DRESSING, SPUNLACE NONWOVEN, 6CMX8CM | 50PCS/BOX, 24BOXES/CTN | 44×27×30 |
| TC1154 | IV CANNULA WOUND DRESSING, PU, 6CMX8CM | 50PCS/BOX, 24BOXES/CTN | 44×27×30 |

Our company is a professional supplier and exporter of medical consumables, lab consumables, and various types of rehabilitation equipment. We pride ourselves on delivering exceptional quality, professional services, and competitive prices in the global market.
We have successfully obtained the ISO13485: 2016 Quality System Certifications and our products are certified by CE European standards. Our products are distributed to over 50 countries and regions worldwide.





Different items have different requirements. Generally, the minimum order value is no less than 2000 USD, though sample orders can be discussed.
Most medical consumable samples can be provided for free. However, as a standard policy, sample freight is collected from the buyer.
Contact us online or send an enquiry with item names and specifications. Once details are negotiated, a 30% TT pre-payment is required to start production.
Yes, we accept OEM, ODM, and OBM orders. We also provide professional packaging design solutions for our clients.
Our facility is ISO13485:2016 certified, and our products carry the CE European certification for safety and quality assurance.
We have a strong production capacity of approximately 5,000,000 pieces per month to ensure timely delivery for large orders.